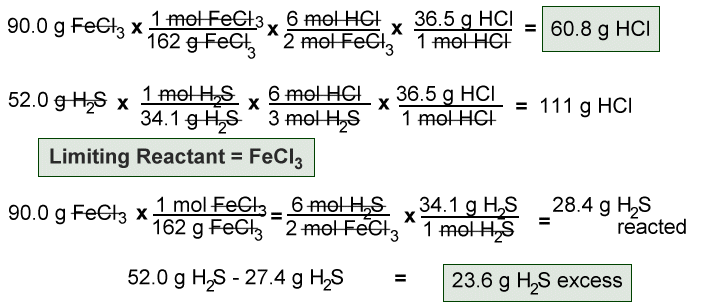What is a Limiting Reactant?

Also known as the limiting reagent is a substance in a chemical reaction which is totally consumed when the chemical reaction is complete. The limiting reactant also determines how much product is formed.
Steps in approaching a limiting reactant problem
- Convert mass of each reactant into moles of each product
- The limiting reactant is the one that produces the least product so determine which reactant produces the lease product
- Calculate the mass of product produced
- Calculate the moles of excess reactant
- Calculate the mass of the excess reactant
Example
- 90.0 g of FeCl3 reacts with 52.0 g of H2S. What is the limiting reactant? What is the mass of HCl produced? What mass of excess reactant remains after the reaction?